Science & Engineering
PIONEERING EXPERTS
Over 99% accurate test in 20 minutes
A game-changing team of medical, technology and operational experts came together in 2019 to pursue a new breed of low cost disease diagnostics deployable anywhere in the field, by anyone.
Collaborating with three British leading molecular biotech universities – Brunel University London, University of Surrey and Lancaster University – our purpose is to develop technologies to solve real world infectious disease challenges and help the control of disease, on the ground, globally.
Our range of scalable testing technologies and products are simple to use, fast, accurate and secure, enabling people everywhere to take more control of health and wellbeing for their communities and stakeholders.
"Human populations are growing and virus and disease threats will always be with us. We are also dependent on and responsible for animals – their health and wellbeing, now and for future generations. Hence the Vidiia team of virologists, clinicians, veterinarians, engineers and software developers have a mission to create affordable and accurate solutions to protect people and communities, everywhere. "
David Rimer, CEO VIDIIA Ltd.
Overview of the test process
Fast, accurate, secure and low cost
Test Comparison
Molecular testing and Lateral Flow tests

VH COVID-19
Reverse Transcription Loop-mediated Isothermal Amplification (RT-LAMP)
- A molecular based test which detects the genetic viral material from a nasal/throat swab sample
- Viral RNA is reversed transcribed into DNA that is then amplified using specific loop primers, reagents and single heating step
- Excellent specificity and Sensitivity, able to detect very low viral load
- Unexpensive equipment and minimal infrastructure
- It can deliver results in minutes

Quantitative Reverse Transcription Polymerase Chain Reaction (RT-qPCR)
- A molecular based test which detects the genetic viral material from a nasal/throat swab sample
- Viral RNA is reversed transcribed into DNA that is then amplified using specific primers and probes, reagents and heating cycles
- Excellent specificity and Sensitivity, able to detect very low viral load
- Requires expensive equipment and infrastructure
- It can deliver results within hours
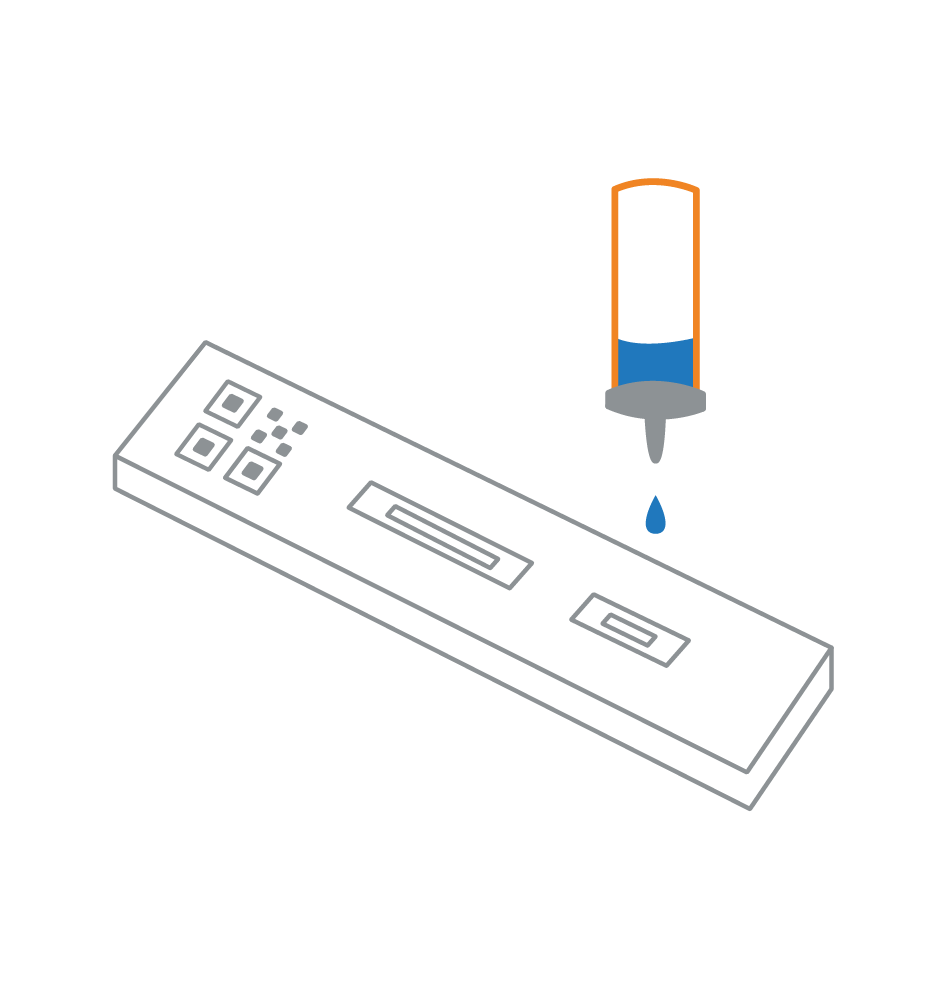
Antigen-Lateral Flow Test (LFT)
- An antigen test which detects the spike protein at the surface of the virus from a nasal/throat swab sample
- Viral protein is detected via an immunoassay
- No amplification, less sensitive, only detects high viral load samples
- No equipment required
- It can deliver results in minutes
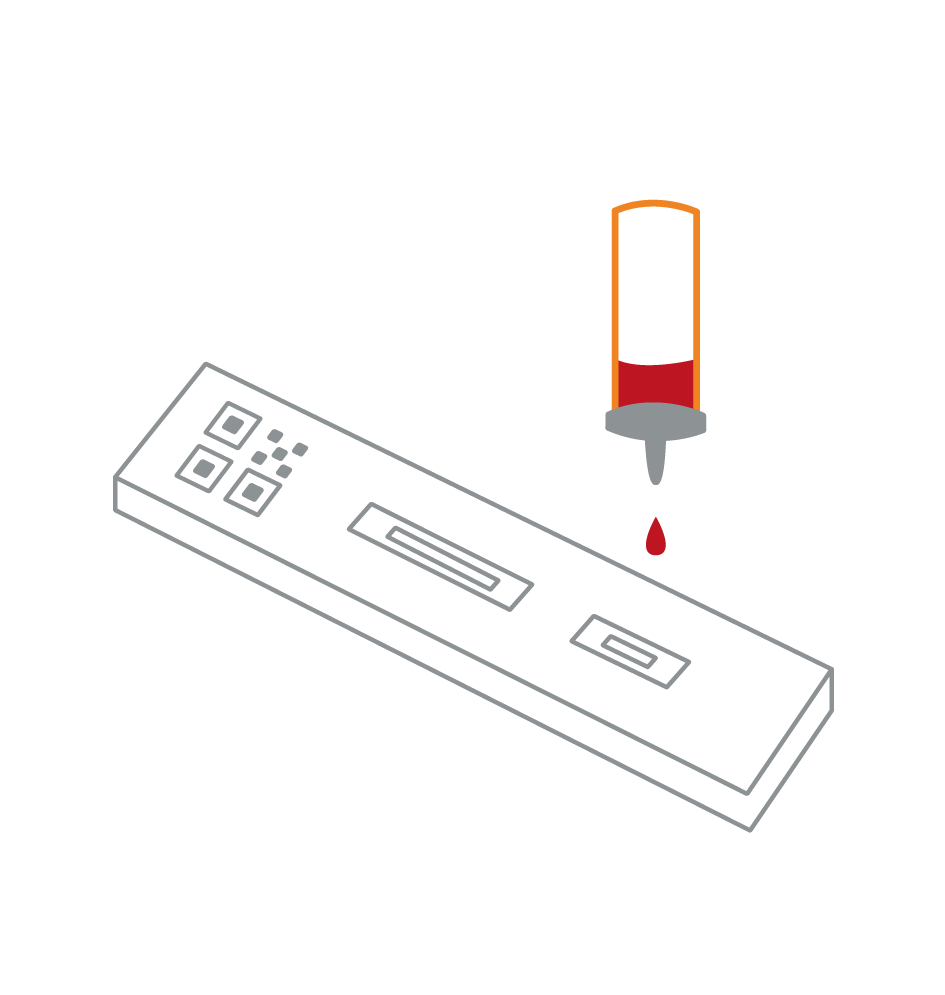
Serological-Lateral Flow Test (LFT)
- An antigen test which detects the presence of antibodies against the virus from a blood sample
- Antibodies are detected via an immunoassay
- Only detects presence of an immune response to a previous viral infection
- No equipment required
- It can deliver results in minutes
VIdiia Hunter
Tailored Software-platform for testing
VIDIIA HUNTER is a platform specifically developed to provide an end-to-end solution for testing; covering all the different requirements for testing; from "Patient" onboarding to results.
Once a "Patient" has been onboarded, the operator can use a mobile app to log the samples and assign them a test position into the VH COVID-19 test. Once the test has started, the VIDIIA VH6 device, powered with Internet of Things (IOT) technology, will take images of each sample and its amplification during the test. Embedded AI machine learning will interpretate the results on behalf of the operator, refining the diagnostics and improving test accuracy and speed.
Finally additional reporting and data analytics is made available in the cloud.
Key Performance Indicators
During a robust clinical study in collaboration with Royal Berkshire County Hospital and Lancaster Infirmary and in partnership with University of Surrey and Lancaster University we successfully collected over 150 positive and 250 negative clinical swab samples for testing using the VIDIIA VH6. All testing was benchmarked against RT-qPCR . This study produced a sensitivity of 99.1% and specificity of 99.6%.
Limit of detection
The VIDIIA Hunter can detect as low as 3 copies of Covid-19 RNA per microliter
Variants of Concern in the UK August 2021
VIDIIA Hunter has been proven able to detect the Alpha, Beta, Gamma, Delta and Omicron variants